Aliri’s Results for the Oligonucleotide Ring Trial Prove Hybridization LC-MS Approach Superior to LBA in Achieving Lower Detection Limits and Higher Specificity
December 2, 2025 – Aliri Bioanalysis, a leading provider of precision bioanalytical and spatial multi-omics solutions for drug developers, recently participated in the Oligonucleotide Ring Trial, in which 10 labs joined together to compare the effectiveness of LC-MS, LBA, and qPCR in quantifying concentrations of oligonucleotides in biological samples. This first-of-its-kind study focused on three types of oligonucleotides, an ASO (Fomivirsen), a GalNAc-siRNA (Lumasiran), and a PMO (Viltolarsen).
Troy Voelker, Sr. Lab Director at Aliri and Chair of an industry leading, multi-lab Oligonucleotide Discussion Group, led the LC-MS method development of the PMO (Viltolarsen), which was analyzed using three mass spectrometry platforms: a QExactive, a time-of-flight (TOF), and a triple quadrupole instrument.
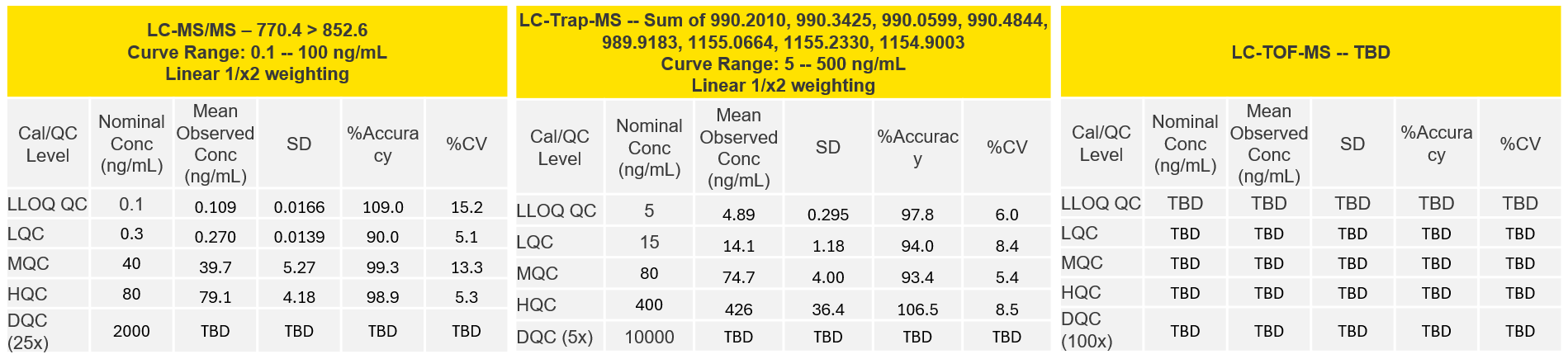
Aliri’s LC-MS hybridization approach achieved detection limits that surpass the specificity and sensitivity of LBA:
- Achieved lower sensitivity (0.1 ng/mL LLOQ) on the triple quadrupole
- Established higher specificity on all LC/MS platforms
- Demonstrated no interference from metabolites on all LC/MS platforms
- Attained unmatched accuracy on the QExactive
- Achieved wide dynamic range & high throughput on all LC/MS platforms
- TOF data coming soon – Stay tuned for more exciting results!
Viltolarsen Platform Analytical Ranges in Rat Plasma
These results are revolutionary for drug developers. Hybridization LC-MS provides the sub-nanogram LLOQs traditionally achievable only with ligand-binding assays, while offering enhanced specificity, structural selectivity, and confirmatory information inherent to mass spectrometric detection. As a result, pharmaceutical programs are no longer restricted to LBA-only workflows when ultra-sensitive PK data are required. With shorter method development timelines than LBA (only 3 weeks compared to 3 months), great accuracy, and wide dynamic range, this method gives drug developers higher confidence than ever in the data they rely on to make critical decisions about the future of their program.

Troy shares,
“It’s exciting that drug developers now have clear data confirming the advantages of hybridization LC–MS over LBA for the bioanalysis of therapeutic oligonucleotides.
I’m looking forward to seeing the industry trend toward greater adoption of this highly effective strategy and proud of the unmatched leadership that Aliri offers customers in this area.”
In the coming months, the Aliri team is eager to release additional research for the Viltolarsen study, which will confirm whether TOF data demonstrates the same sensitivity observed on our triple quadruple and just how low qPCR will reach for concentration sensitivity.
To view the full results of Aliri’s hybridization LC-MS study for the Oligonucleotide Ring Trial, download Troy Voelker’s recent presentation here.